CERTAINTY IS OUR COMMITMENT
CERTAINTY IN EVERY BUILD - TRUSTED AROUND THE WORLD
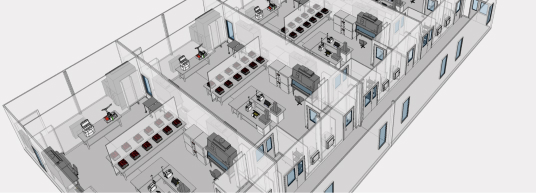
Companies around the world trust G-CON to deliver advanced cleanroom solutions for PODular and hybrid construction options. As a trusted cleanroom manufacturer, G-CON specializes in innovative cleanroom manufacturing solutions that accelerate pharmaceutical and biotech facility builds worldwide. From prefabricated POD® Cleanrooms to modular cleanroom components, we deliver scalable, sustainable environments for critical manufacturing processes.
Why Leading Companies Trust G-CON for Cleanroom Manufacturing Excellence
G-CON’s expertise in cleanroom manufacturing is unmatched. As a premier cleanroom manufacturer, we combine cutting-edge design with rapid, reliable delivery—ensuring clients meet evolving demands without compromising on quality or compliance.
Learn More About G-CON

RELIABLE SPEED
ACCELERATE YOUR FACILITY BUILD WITH TRAILBLAZING SPEED & EFFICIENCY
PODular, prefabricated designs allow for rapid delivery to meet changing capacity demands. POD® based facilities can be implemented and ready to use in as little as 6 months.
Explore Cleanroom Products

UNINTERRUPED SCALABILITY
SCALE UP AND SCALE OUT EXISTING PROCESSES
POD® Cleanrooms allow customers to ramp-up quickly if drug demand increases with minimal impact to existing operations when new units installed for scale out.
Get a QuoteA PROVEN PROCESS
Cleanroom Manufacturing Solutions from the Global Leader in Prefabricated Cleanrooms

G-CON's proven POD delivery model includes standardized methods and procedures covering every aspect of a project from design through qualification, all controlled under its ISO 9001 certified Integrated Management System (IMS). The G-CON team is the most highly trained and experienced in delivering pre-fabricated cleanroom solutions