Feb 03, 2026
Flexible Cleanroom Design: How Prefabricated Cleanrooms Enable Scalability
The pharmaceutical and biotech industries are facing increasing uncertainty in demand, due to diverse product pipelines, accelerated clinical trials and evolving regulatory expectations. Traditional stick-build cleanroom construction often locks companies into rigid designs that cannot easily adapt to these dynamic requirements.
Flexible cleanroom design, enabled by prefabricated cleanroom PODs, provides a fundamentally different approach: they offer flexibility by design, enabling organizations to rapidly configure, repurpose, or expand cleanroom capacity in response to shifting needs. This inherent flexibility translates into scalability, whether that means adding new suites for a growing portfolio, adjusting classifications for different modalities, or replicating facilities globally.
Why Flexibility Is Critical in Pharmaceutical and Biotech Cleanrooms
Pharmaceutical and biotech industries no longer operate in a static manufacturing environment. Emerging therapies such as cell and gene therapy, personalized medicine, and novel biologics require cleanrooms that can adapt as pipelines evolve. A rigid, one-time buildout will either risk under-capacity or unusable space depending on how modalities and therapies evolve.
Flexible cleanroom design addresses these challenges by:
- Configurable layouts: PODs can be designed for different classifications and are reconfigurable as requirements shift
- Modular workflows: Facilities can be adapted to support different unit operations without extensive reconstruction
- Repurposing capability: PODs can be relocated or reassigned to new products, supporting lifecycle changes and multi-product strategies
Key Benefits of Flexible Cleanroom Design
Flexibility in a cleanroom design is not just about short-term convenience but long-term resilience in facility strategy.
While scalability is a critical driver for any facility design, true scalability is only possible if the cleanroom design is flexible. Prefabricated PODs deliver this through standardized yet configurable units that can be deployed in a phased / scale-out manner.
Key advantages include:
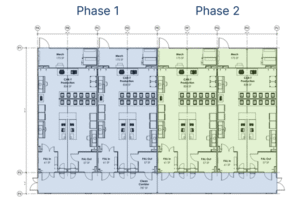
- Scale-out: Additional PODs can be added incrementally as demand grows, reducing the need for oversized upfront investments. Refer to Figure 1.
- Scale-up: PODs can be designed to support higher volumes or accommodate new equipment, enabling increased throughput without major facility disruption.
- Multi-site replication: Standardized POD designs allow companies to replicate facilities globally, ensuring consistent quality and operational models.
The clonability of the PODs reduces both technical risk and capital exposure, while ensuring facilities remain aligned with actual demand.
Figure 1: Scale-out strategy using prefabricated cleanroom PODs
By adopting a POD-based strategy, companies can reduce risk, accelerate time-to-market, and future-proof their manufacturing footprint against the uncertainties of modern drug development. Designing a cleanroom to be flexible and planning for scalability allows pharmaceutical and biotech industries to derisk future uncertainty and flexible cleanroom design using prefabricated solutions is one proven way to address these evolving challenges.